Yahoomagazine
Spettacolo

Maria De Filippi perde le staffe. Lo sfogo contro Gianni Sperti: “Ma sei serio?”
Maria De Filippi perde le staffe contro l’opinionista Gianni Sperti: “Ma sei serio?”. Lo sfogo …

Mirko Brunetti e Perla Vatiero, come vanno i rapporti fuori dalla casa. Lui ammette: “Non riuscivamo a vederci e…”
Mirko Brunetti e Perla Vatiero, come vanno i rapporti ora che il “Grande Fratello” è …
News

Chiari, dramma nel pomeriggio: 45enne trovato senza vita in strada da un passante
Un uomo di 45 anni è stato trovato morto in strada da un passante: la …

Napoli, incidente nella Galleria Laziale: perde la vita un 37enne
Un giovane di 37 anni è morto questa mattina in un tragico incidente stradale verificatosi …
Tecnologia

Nuovo aggiornamento Telegram: se lo scaricate non tornerete più indietro
Gli sviluppatori di Telegram hanno introdotto una fantastica novità per gli utenti: tutto quello che …

Apple lancia l’avviso agli utenti: “iPhone spiati da uno spyware mercenario in 92 nazioni”
Apple presa d’assalto: gli iPhone di mezzo mondo attaccati da uno spyware mercenario. Cosa fare …

Whatsapp, ora puoi mettere musica e suoni su una foto
Arriva una novità clamorosa per Whatsapp e le foto: ora è possibile aggiungere anche musica …
Musica

Il Volo, info e prezzi della data estiva di Marsciano
Sono diverse le date inserite nel tour “Tutti per uno – Capolavoro” de Il Volo. …

Mare Fuori, arriva la pesante bocciatura: “Non è adatta al..” Annuncio sconcertante
E’ in arrivo una bella stroncatura per la fiction di Mare Fuori: pare, infatti, che …
Economia

D’ora in poi è obbligatorio inserire anche questa spesa nella Dichiarazione dei Redditi
È tempo di dichiarazione dei redditi; ma quali sono le spese che vanno effettivamente segnalate …

La lista dei consumi elettrodomestico per elettrodomestico
Vuoi conoscere i tuoi consumi? Vediamo insieme l’impatto che hanno i vari elettrodomestici: pazzesco tutto …
Lifestyle

Piano cottura ad induzione: occhi aperti, se per sbaglio lo dimentichi acceso succede questo
Se stai pensando di installare nella tua cucina un piano cottura ad induzione, oppure lo …

Viaggiare con un solo bagaglio a mano, parlano le hostess: “6 errori che tutti i passeggeri commettono”
Viaggiare con il bagaglio a mano impone regole stringenti: quali sono gli errori più frequenti …
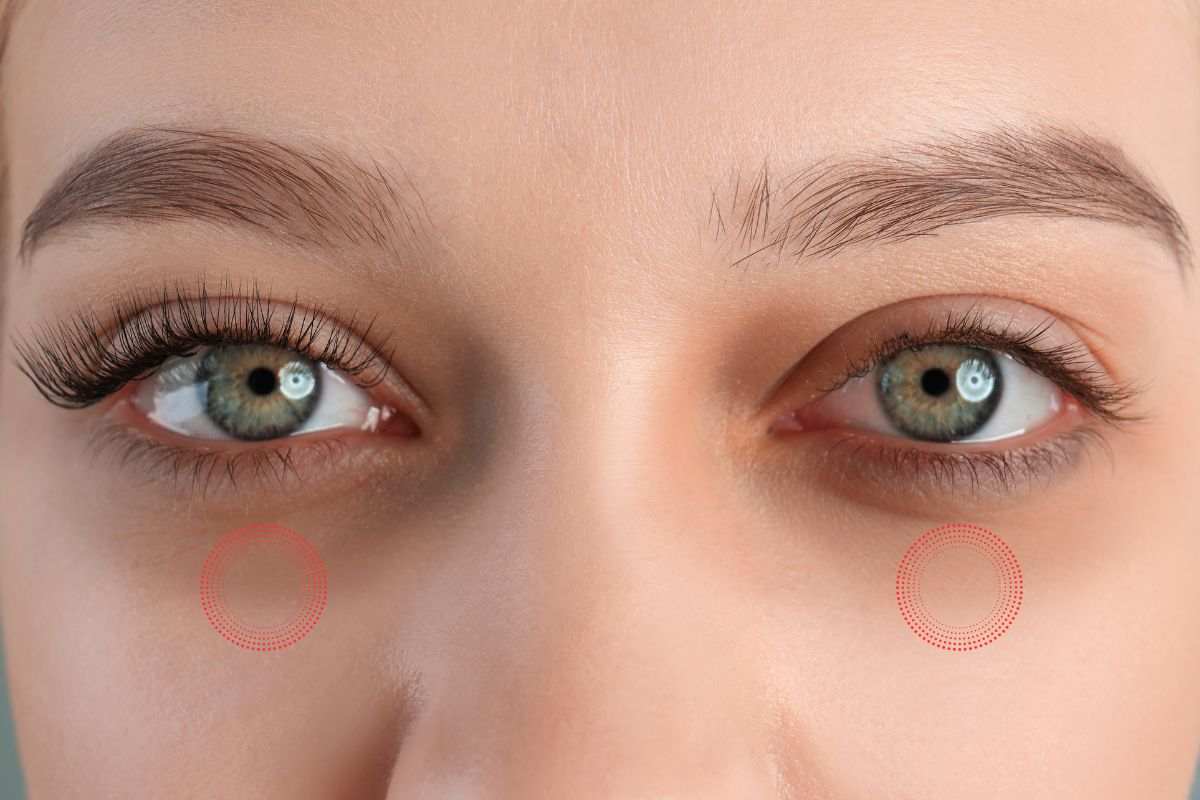
Occhiaie e sguardo spento? La soluzione sta dal fruttivendolo: non lo avresti mai pensato
La primavera comporta tanta stanchezza, sonnolenza, sguardo spento e occhiaie: dal fruttivendolo troviamo la soluzione. …
Sport

Gambe toniche ma non gonfie, fai attenzione a questi errori: possono fare la differenza
Gambe toniche ma non gonfie, come riuscire ad ottenerle: fai attenzione a dei piccoli errori …

Sinner, la sconfitta che lo rende vincitore nato
Il ko di Monte Carlo avrebbe potuto essere evitato se Sinner avesse richiesto l’intervento dell’arbitro. …

Spagna, muore giovane promessa della ginnastica: stroncata a 17 anni da una meningite
Ieri, una giovane atleta spagnola di 17 anni è morta in ospedale, dove era arrivata …
